You are here: Home: CCU 5 | 2004: James Cassidy, MD, MBChB, MSc, FRCP
| James Cassidy, MD, MBChB, MSc, FRCP |
EDITED COMMENTS |
 The Xeloda® in Adjuvant Colon Cancer Therapy trial The Xeloda® in Adjuvant Colon Cancer Therapy trial
Trial design
In the Xeloda® in Adjuvant Colon Cancer Therapy (X-ACT) trial, the Mayo Clinic regimen was chosen as the standard treatment because: (1) it was the predominant schedule used in North America, (2) it was the regimen preferred by the FDA as a comparator, and (3) analyses conducted in European countries indicated that the bolus regimen was still frequently used. Capecitabine was selected as the other treatment. Approximately 2,000 patients with Dukes’ C colon cancer were randomly assigned within eight weeks of their surgery to the six courses of Mayo Clinic regimen or eight courses of capecitabine (2.1).
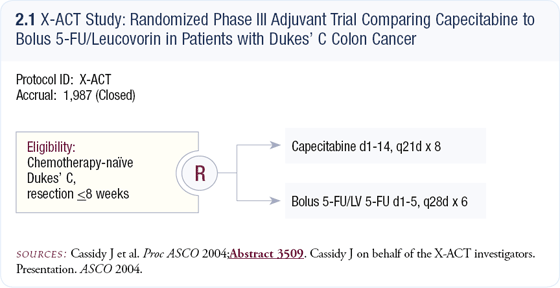
Demographics
Patients over the age of 75 were not permitted in the original protocol; however, a number of protocol waivers allowed patients older than 75 years to be entered into the trial. The age range of the patients in the trial reflects a disease that occurs predominantly in patients in their sixties and seventies. Our data are comparable to data from any other clinical trial. We’ve deliberately analyzed what happens to older patients within the protocol, and no toxicity or efficacy disadvantages are associated with patients older than 75 years.
Efficacy
The trial was powered to show equivalence in the primary endpoint — diseasefree survival (DFS). A number of secondary endpoints were relapse-free survival (RFS), overall survival (OS), pharmacoeconomics and quality of life. The patients in the X-ACT trial have been followed for a median of 3.8 years. In terms of the primary endpoint of DFS, the trial demonstrated equivalence between the two treatments.
A statistically significant difference was observed in RFS; fewer patients treated with capecitabine experienced a relapse in their disease. Overall survival was also improved for patients treated with capecitabine, but it was not a statistically significant improvement. Because the trial did not include many patients yet at risk for death, we must wait to determine the precise answer about the difference in OS. In conclusion, capecitabine is at least equivalent to the Mayo Clinic regimen and probably better.
Toxicity
Capecitabine was less toxic than the Mayo Clinic regimen in terms of diarrhea, mucositis, neutropenia and neutropenic sepsis. However, more patients treated with capecitabine developed hand-foot syndrome, which is a nuisance and requires dose reductions but is not life threatening. In my opinion, hand-foot syndrome is quite low in rank order of importance. The treatment-related mortality rates were 0.3 percent with capecitabine and 0.4 percent with the Mayo Clinic regimen. The treatment-related deaths were due to a combination of diarrhea and neutropenia. Compared to other adjuvant trials reported in the literature, that mortality rate was lower in the X-ACT trial.
Over 80 percent of the patients in both arms received the full duration of therapy. Just over half of the patients in both arms required a dose reduction, delay or interruption. With capecitabine we have the potential to interrupt the dosing, and patients and caregivers must be involved in that process.
Educating patients about capecitabine-associated side effects
We make an effort to educate patients about the potential for diarrhea because if patients develop diarrhea, they may become dehydrated and require hospitalization. Sometimes diarrhea is associated with neutropenia. Diarrhea and neutropenia together are dreaded side effects of the fluoropyrimidines. Patients must be admitted to the hospital to receive broad-spectrum antibiotics and rehydration. I tell my patients they should stop treatment and inform us if they are having diarrhea more than five times in a 24-hour period.
Hand-foot syndrome is a bit more subtle. Patients often develop a minor degree of hand-foot syndrome with the first cycle of capecitabine, and it may be worse with the second cycle. At that point, we reduce the capecitabine dose. Because of that strategy, I don’t see many patients with severe hand-foot syndrome. We also tell patients to stop treatment if they develop redness of their hands or feet with pain that interrupts their level of functioning.
CALGB-89803: Adjuvant trial comparing IFL to bolus 5-FU/ leucovorin
CALGB-89803 compared IFL to the Roswell Park bolus 5-FU/leucovorin regimen. The Roswell Park regimen is probably equivalent to the Mayo Clinic regimen and may be less toxic. It was a resoundingly negative trial; no differences between the two treatments were found. It has been questioned whether the early deaths encountered with the IFL regimen were sufficient to have negated any possible benefit. The results were unexpected. In patients with advanced disease, IFL seems to be a better regimen than 5-FU/leucovorin; hence, it’s counterintuitive to not have a positive study in the adjuvant setting.
Adjuvant therapy options for patients with Stage III disease
Adjuvant therapy options include the Mayo Clinic regimen, capecitabine or the addition of oxaliplatin to 5-FU/leucovorin (FOLFOX). Based on the X-ACT trial results, the standard of care may deviate from the Mayo Clinic regimen towards single-agent capecitabine. For me, it’s a done deal. Others may say, “It’s only one trial. We need more data.”
Recent trials evaluating celecoxib with chemotherapy
Interesting results from trials evaluating celecoxib in combination with chemotherapy were reported at ASCO 2004. In one trial, celecoxib’s ability to reduce the incidence of oxaliplatin-related neurotoxicity was tested. Concurrent use of celecoxib and oxaliplatin appears to cause less neurotoxicity. In another clinical trial from the French group conducting the OPTIMOX study, in which oxaliplatin is administered, stopped and then reintroduced, that strategy was administered concurrently with celecoxib and the response rates were lower than the response rates historically seen in the OPTIMOX study. I wonder whether a pharmacokinetic or pharmacodynamic interaction between celecoxib and oxaliplatin reduces the activity and toxicity of oxaliplatin.
Researchers from MD Anderson reported trial results suggesting that celecoxib in combination with capecitabine might cause less hand-foot syndrome. I would like to see that evaluated in a larger, more formal trial. I’d also want to ascertain that a reduction in the activity of capecitabine didn’t occur along with the reduction in the incidence of hand-foot syndrome.
Educating patients about oxaliplatin-related neurotoxicity
We have a discussion with patients about short-term neurotoxicity, such as laryngopharyngeal dysesthesia, because it’s quite frightening. If they are not told that it can happen, they become scared when the symptoms occur. Then, some patients may not want to be treated with a second cycle.
If they are educated in advance, they cope with it. In terms of the long-term cumulative neuropathy, we ask: Do you have neuropathy that is persistent between cycles? Does the neuropathy have a functional consequence? If the answer is yes to either of those two questions, then we’ll reduce the oxaliplatin dose.
Select publications
 |
| Dr Cassidy is a Professor of Oncology and Head of the Department of Cancer Research in the UK Department of Medical Oncology at the University of Glasgow in Glasgow, Scotland. |
|

